Earlier this week, the U.S. Food and Drug Administration (FDA) announced that it has approved the commercialization and use of a new drug, one that will target ALL.
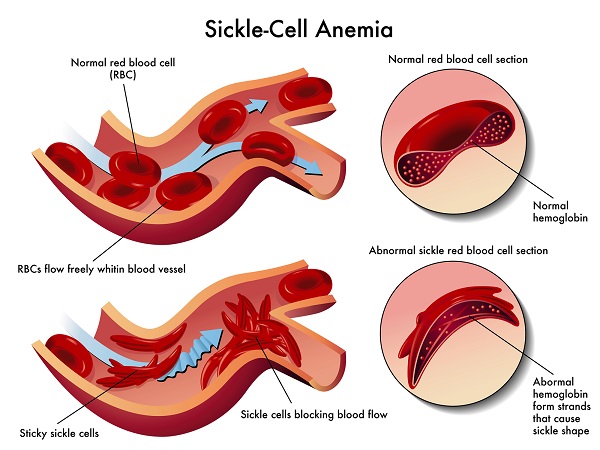
ALL or B-cell precursor acute lymphoblastic leukemia is a rare and rapidly progressing form of blood cancer. It is caused by the bone marrow producing too many B-cell lymphocytes, which are a type of immature blood cells.
According to the National Cancer Institute, almost 6,000 people in the United States will be diagnosed with all this year alone. Some 1,440 will possibly die because of it.
ALL Patients Will Have A New Drug Variant
The FDA approved the use of a new drug for this type of blood cancer, one that can be utilized by adult patients. It was approved for those that tried other treatments that ultimately failed. The medicine can be used by adults with a relapsed or refractory ALL.
“These patients have few treatments available and today’s approval provides a new, targeted treatment option,” stated Richard Pazdur.
An M.D., he is the FDA director of the Oncology Center of Excellence. Dr. Pazdur underlines that patients whose disease returned or did not respond to treatment typically have a low life expectancy.
The newly approved drug is Bespona, and Pfizer Inc. commercializes it. Before receiving the mark of approval, this medicine’s effects were tested in a trial involving almost 220 patients.
Among those given Bespona, 36 percent saw their blood cancer for a period of eight months, on average. Among those to receive chemotherapy, 17 percent saw their disease in complete remission for an average of five months.
This new drug is believed to work by binding to the surface of the cancerous cells and as such, blocking its growth.
The medicine is injectable, but it also carries the most stringent warning from the FDA as it could cause severe adverse reactions.
Image Source: Flickr